What are MIS-C and MIS-A?
MIS-C and MIS-A stands for Multi-Inflammatory Syndrome in Children (MIS-C) and Adults (MIS-A) respectively.
MIS-C and MIS-A present with persistent fever and hyper-inflammation with multi-organ failure (cardiac, gastrointestinal, renal, haematological, skin, and neurological) following SARS CoV2 infection; they are clinically almost indistinguishable from another condition called toxic shock syndrome.
The current diagnostic criteria for MIS-C and MIS-A can be found on the Centre for Disease Control and Prevention (CDC) website. Essentially the diagnostic criteria depend on whether the patient is over or under 21 years old and include:
- Fever > 38 oC for ≥ 24 hours
- 2 organ involvement (cardiac being the most common)
- Laboratory evidence of inflammation e.g. high CRP, Procalcitonin, neutrophils, etc.
- Recent Covid-19 based on a positive lab test e.g. PCR
MIS-C is rare, occurring in approximately 2 in 100,000 children after Covid-19. The incidence of MIS-A is unknown as it often overlaps with severe Covid-19, so it’s not known if it is more common or not in adults than in children.
Despite knowing about MIS-C and MIS-A for over 18 months no one has known why it occurs and why it seems to happen in only a few people and not others. However, an increasing body of evidence from researchers in the US shows that MIS-C and MIS-A are caused by a “superantigen” produced by SARS CoV2… and anything that has “super” in front of it has to be great… right?
What is an antigen and how does it acquire its “super powers”?
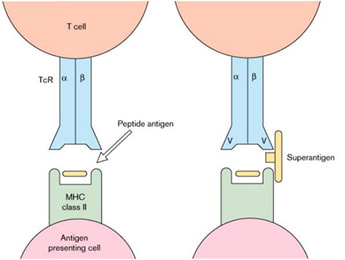
An antigen is a molecule that can bind to an antibody or T cell receptor. They are a form of “communication” and the way the immune system “recognises” that something is happening. Antigens can be floating around in fluids such as the blood stream or can be attached to cells; some antigens are specifically produced by infected cells and then expressed, in something called a Major Histocompatibility Complex (MHC) receptor on a cell surface in such a way that helps the T lymphocyte cells recognise that the antigen is important and that they need to start to produce an immune response against it.
Most of the time antigens in MHC receptors stimulate a tiny fraction of the total T cells in the body, so the immune response starts to mount slowly overtime therefore it does not cause a massive overreaction from the immune system, which would harm the person.
However, “superantigens” can bind to lots of MHC receptors of many cells. This lack of choosiness means they trigger a massive response from T cells; a person can activate about 20-30% of their T cells at the same time. This causes a massive overreaction from the immune system damaging lots of different organs in the body. Needless to say then, superantigens are not a good thing!!! Some super “heroes” are villains…!!
Okay, I admit it, I’m a geek, but this is the bit I find fascinating!
Conventional teaching in microbiology and medicine told us that it is only bacteria that produce superantigens, such as toxic shock syndromes from Staphylococcus aureus producing Toxic Shock Syndrome Toxin (TSST) or Staphylococcal Enterotoxin A or B (SEA or SEB). But when you stop and think about it, it doesn’t have to just be bacterial; any protein could be a superantigen. Viral replication leads to new proteins just as readily as with bacteria. The new protein just has to be capable of activating vast amounts of the innate immune system.
So, it turns out that replication of SARS CoV2 can indeed produce a superantigen. Some amazing work by a group of researchers in the US has discovered that the S1 fragment of the SARS CoV2 spike protein has the same “protein structure” as Staphylococcal Enterotoxin B; this “protein structure” is mimicking what we would normally consider a bacterial superantigen, it’s like Pepsi cola looking or tasting like Coca-Cola. The same researchers have shown that not only is the structure the same but the S1 protein fragment is functioning in the same way as a “bacterial superantigen” and can cause a multi-inflammatory syndrome in animal studies. And then, to put the icing and cherry firmly on the top of their research cake, the researchers showed that blocking the S1 fragment superantigen prevented the multi-inflammatory syndrome in their animals! Brilliant, just brilliant!!!!
Why doesn’t everyone with Covid-19 get “attacked” by the “super villain” MIS-A and MIS-C?
Wait a minute! I hear you cry… if SARS CoV2 produces a superantigen why doesn’t everyone get MIS-C or MIS-A?
Well, there are two reasons why not everyone exposed to a superantigen will react to it:
- Most older children and adults have antibodies against Staphylococcal Enterotoxin B – we are exposed to lots of bacteria and proteins during our early years and by the age of 16 years about 80% of the population have antibodies against SEB and so are likely to be immune to developing the multi-inflammatory syndrome. However, as we move into old age our immune systems become less efficient and many of us lose our ability to produce these anti-SEB antibodies. This may partially explain why we see more of the multi-inflammatory syndrome in children and the elderly.
- There is a genetic predisposition to being able to react to SEB – in order to react to SEB you have to have specific MHC receptors on you cell surface, and these are not common. If you don’t have the right MHC receptor the superantigen doesn’t bind and therefore doesn’t activate the extreme immune response. The specific MHC receptors that predispose to the SEB reaction are more common in people of African or Hispanic ethnic origin; the multi-inflammatory syndromes are more common in people of these ethnic origins. It turns out that the specific MHC receptors needed do not occur in people of Asian ethnic origin, which probably explains why MIS-C and MIS-A have not been seen in China, South Korea or Japan.
Why do MIS-C and MIS-A not occur during the acute infection?
This is a tricky one. MIS-C and MIS-A usually occur weeks after the acute Covid-19 infection; if it is all down to spike protein why doesn’t it occur during the acute infection when there is plenty of spike protein around?
This hasn’t been completely explained, but the theory is that some people have long term production of viral proteins even after the infection has settled. Children in particular seem to have prolonged production of proteins in the gastrointestinal tract after Covid-19 (being in the gastrointestinal tract isn’t that surprising, as many coronaviruses are actually gastrointestinal pathogens). It is thought that this prolonged production of proteins, in particular the S1 fragment of the spike protein, is what triggers the superantigen response. However, this is still a bit woolly for me; I think there is more work to be done on this area….
How do we explain the reduction in MIS-C and MIS-A with the emergence of the Delta variant of SARS CoV2?
As the Delta variant of SARS CoV2 became the dominant version of SARS CoV2 (well dominant before Omicron came along!) the incidence of MIS-C and MIS-A dropped dramatically but severe Covid-19 and hospital admissions in children and younger adults became more common! Why would this happen? This was a bit odd as it was unclear why a small change in virus variant would lead to such a dramatic change in the way Covid-19 would present in a different age group; it probably wasn’t just that younger people weren’t vaccinated. The superantigen related research does go some way to explaining the clinical observations.
The Delta variant of SARS CoV2 changed its spike protein which led to higher rates of cleavage, or breakage, of the spike protein. This in turn led to more virus and more spike protein fragments. There is experimental evidence that this increase in spike protein “fragments” causes more severe early Covid-19 in all age groups, including young people, and because more of these young people are getting severe early Covid-19 they are developing an immune response before they are exposed to the later superantigen effects. Bizarrely, early severe Covid-19 seems to prevent, or protect from, superantigen MIS-C and MIS-A.
How does knowing this help in treating MIS-C and MIS-A?
Okay, so what? How does knowing all of this stuff help us in managing patients with MIS-C and MIS-A?
Well, if we know that MIS-C and MIS-A are essentially a superantigen response in the same way as toxic shock syndrome from S. aureus for example, then the treatment should be the same. And guess what? It is….
Patients with MIS-C and MIS-A respond really well to intravenous immunoglobulin (IVIg), in addition to the usual steroids and immunomodulators which dampen down the excessive immune response. It puzzled me that IVIg made such a difference to MIS-C and MIS-A because IVIg is made from the blood donations of hundreds of people and contains antibodies from the different types of antigens these donors have come across in their lifetimes. But, as Covid-19 is such a new virus the amount of anti-SARS CoV2 antibody in IVIg might be expected to be low and in addition the antibody would be against various viral proteins. It is hard to see how this would damp down an immune response AFTER the infection has already occurred; added to this there is a lot of evidence that antibodies against SARS CoV2 make little difference in most people if given after the first few days of infection. And yet IVIg works against MIS-C and MIS-A.
Now, the benefit of IVIg in MIS-C and MIS-A makes a lot more sense IF the antibody in the IVIg ISN’T specifically against SARS CoV2 BUT actually against a superantigen that is driving the excessive immune response. So, if we assume 80% of donors have antibody against SEB in levels that protect these donors against SEB, then when we combine the hundreds of donor samples into IVIg we get vast quantities of anti-SEB antibody in IVIg. The anti-SEB antibody recognises and neutralises the spike protein S1 fragment superantigen because it is essentially the same “protein structure”. There is a beneficial cross-reacting antibody that takes out the “villainous” superantigen in a process of false recognition! How lucky is that!! I think that is so cool… but then as I’ve already admitted, I am a bit of a geek….
What about vaccines based on spike protein?
This is an important question; if vaccines are based upon the spike protein of SARS CoV2 and the S1 fragment of the spike protein is the superantigen, do the vaccines cause MIS-C or MIS-A? Well, we don’t know about all vaccines but apparently the Johnson & Johnson and Novavax vaccines have a modified spike protein. It seems that the sequence of RNA that causes the superantigen S1 fragment to be cleaved off in very high amounts has been removed from the vaccine sequence as it made the vaccine less stable and less able to promote a good immune response. It seems that by luck a problem with vaccine instability led the vaccines creators to remove the potential for the superantigen response.
I haven’t been able to find out if the same is true for the other Covid-19 vaccines like the Pfizer and Astra Zeneca vaccines, but it does seem that the multi-inflammatory syndrome has been seen after the Pfizer vaccine. It’s VERY important to note though that the multi-inflammatory syndrome is considerably rarer than severe Covid-19 in the unvaccinated! Vaccines are still important!!
What else might the protein similarity evidence explain?
Well, it turns out that the S1 fragment superantigen isn’t the only molecular mimicry going on in Covid-19. There are also sequences of protein in SARS CoV2 that are able to produce proteins that resemble certain neurotoxins which have high affinity for other MHC receptors. These proteins are known to have the potential to cause encephalitis and chronic neurological symptoms reminiscent of long Covid.
In fact, I heard the US researchers who have studied the superantigen in spike protein talk about a research paper from 1982 called “Persistent neuropsychological sequelae of toxic shock syndrome” by Rosene, Copass, Kastner, et al, which could be describing long-Covid rather than toxic shock syndrome. It could be coincidence, but if this is the case then it may help point the way to more effective treatments of long Covid using immunomodulators and possibly IVIg.
So, there we have it. Everything I knew about bacterial superantigens does in fact apply to Covid-19. Just when I thought Covid-19 was getting really dull and boring something comes along to kick me up the bum and remind me that Covid-19 still has some surprises left in store. Rats! Just when I thought I had all things Covid-19 sussed….


 RSS Feed
RSS Feed
