Well, sometimes it isn’t as simple as that. Sometimes you need more than just a standard laboratory investigation… you need to take your antibiotic testing to another level… do you mean levitation?!
These questions came up this week with a particularly tricky infection. The patient has endocarditis, but the bacterium is unusual and therefore there are no guidelines to tell us how to treat them and there are very few case reports on “Dr Google” where other people have tried to treat this type of infection before. There isn’t really anyone else with experience of treating this, so what can you do? Levitation?!
No, in this situation we have to go back to basics and really work out how sensitive this bacterium is to the antibiotics we want to give.
But first, we need to look at how we normally test antibiotic susceptibility in the microbiology laboratory?
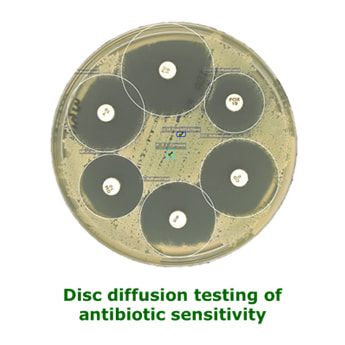
Disc diffusion
Most microbiology laboratories in the UK use disc diffusion to investigate whether a bacterium is likely to respond to treatment with a particular antibiotic. Labs use standard operating procedures (SOPS) which are usually based on a published method of testing, and which are standardised and repeatable across labs e.g. EUCAST (European Committee on Antimicrobial Susceptibility Testing).
The method involves a broth of bacterium being spread all over the agar plate and then a filter paper impregnated disc being placed on the agar plate. The plate is then incubated to allow the bacterium to grow. If the bacterial growth is inhibited by the antibiotic the bacterium will not grow and the agar will be clear.
Results are reported as resistant, susceptible and high level susceptible (R, S and S* see earlier Susceptibility Testing blog) and are based on the measured diameter of the “zone of inhibition” or clear zone around the antibiotic disc; this diameter depends on the concentration of the antibiotic in the agar as it diffuses away from the antibiotic disc… the further away from the disc the lower the concentration.
- The zone size cut off is based on a standard dose of antibiotic given to a 70kg person – higher doses of antibiotics may still work even if the result says resistant, likewise morbidly obese patients may not respond to treatment even though the result says susceptible
- The result is essentially an all or nothing result, resistant or susceptible – there is no detail as to “how” susceptible the bacterium is, it may only just be susceptible or it may be incredibly susceptible, disc diffusion methods do not give that level of detail. This matters if a bacterium is just susceptible and perhaps the patient is obese or misses a dose of antibiotic, they will quickly become subtherapeutic and may not be treated adequately
- You can only test antibiotic/bacterium combinations for which there is a method –the EUCAST method has lots of options for antibiotics against commonly isolated bacteria, but it is not infallible. Rare bacteria or unusual antibiotic/bacterial combinations are not covered in the method, and yet you may be treating something rare or having to use suboptimal antibiotics because of allergies or complications
So, what do you do when there isn’t a disc diffusion option for testing antibiotic susceptibility? Levitation?! No, MIC…
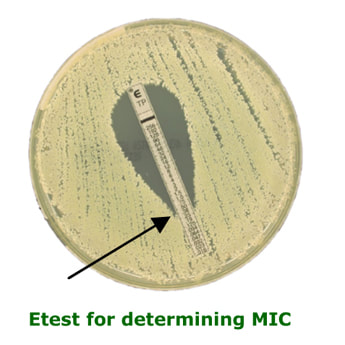
Minimum Inhibitory Concentration (MIC)
If you want to know more detail about just how sensitive a bacterium might be to an antibiotic you can work out something called the Minimum Inhibitory Concentration (MIC).
The MIC is the least amount of antibiotic required to prevent a bacterium from multiplying. The bacterium may still be alive. It is only usually performed in specific clinical scenarios under the instruction of a Microbiologist, e.g. infective endocarditis. The most common method employed in most UK laboratories is the Etest method whereby a strip impregnated with an antibiotic gradient is placed on an inoculated agar plate. The MIC is determined by how far up the strip the bacterium can grow. Low concentrations (bottom of strip) allow growth whereas higher concentrations (top of strip) inhibit the growth. The MIC is the point at which the growth meets the strip.
- Compare the MIC to a standardised method for treating an infection e.g. endocarditis guidelines, and use the guideline to tell you how to treat the patient
- Compare the MIC to the measured levels of antibiotic in the patient’s blood, from therapeutic drug monitoring, to see if there is enough antibiotic present to inhibit the growth of the bacterium
So, the MIC is great and it is usually enough to help you decide how to treat the more complicated patients but sometimes you have to go even further… welcome to the bacterial term “tolerance”…
What is microbiological tolerance?
No, microbiological tolerance is not what the Microbiologist has to show when someone rings up and asks a daft question! It is generally “known” that Microbiologists lose tolerance over the day!
Microbiological tolerance is the difference between the amount of antibiotic required to inhibit the growth of a bacterium (MIC) and the amount required to kill the bacterium (known as the Minimum Bactericidal Concentration or MBC).
For most bacteria the tolerance is relatively low, that is the difference between MIC and MBC is small. Putting it another way, you don’t need to increase the concentration by much to go from inhibiting to killing the bacterium e.g. Streptococcus pneumoniae has low tolerance to beta-lactams. Other bacteria have high tolerance; the amount of antibiotic required to kill the bacterium is much higher than that required to inhibit the growth e.g. Enterococcus faecalis has high tolerance to beta-lactams.
Remember, most of the time this doesn’t matter, we know tolerance exists and take it into account for complex infections caused by common bacteria e.g. simple infections such as catheter associated urinary tract infections due to Enterococcus faecalis can be treated with PO Amoxicillin 1g TDS (we only need to inhibit the bacterium while we remove the catheter and the immune system clears the bacterium). Whereas infective endocarditis caused by the same bacterium requires IV Amoxicillin 2g 4 hourly (12g per day!) in order to give enough antibiotic to kill the Enterococcus faecalis, the immune system won’t clear this infection on its own due to the infection’s location.
So how do we work out the MBC for a bacterium?
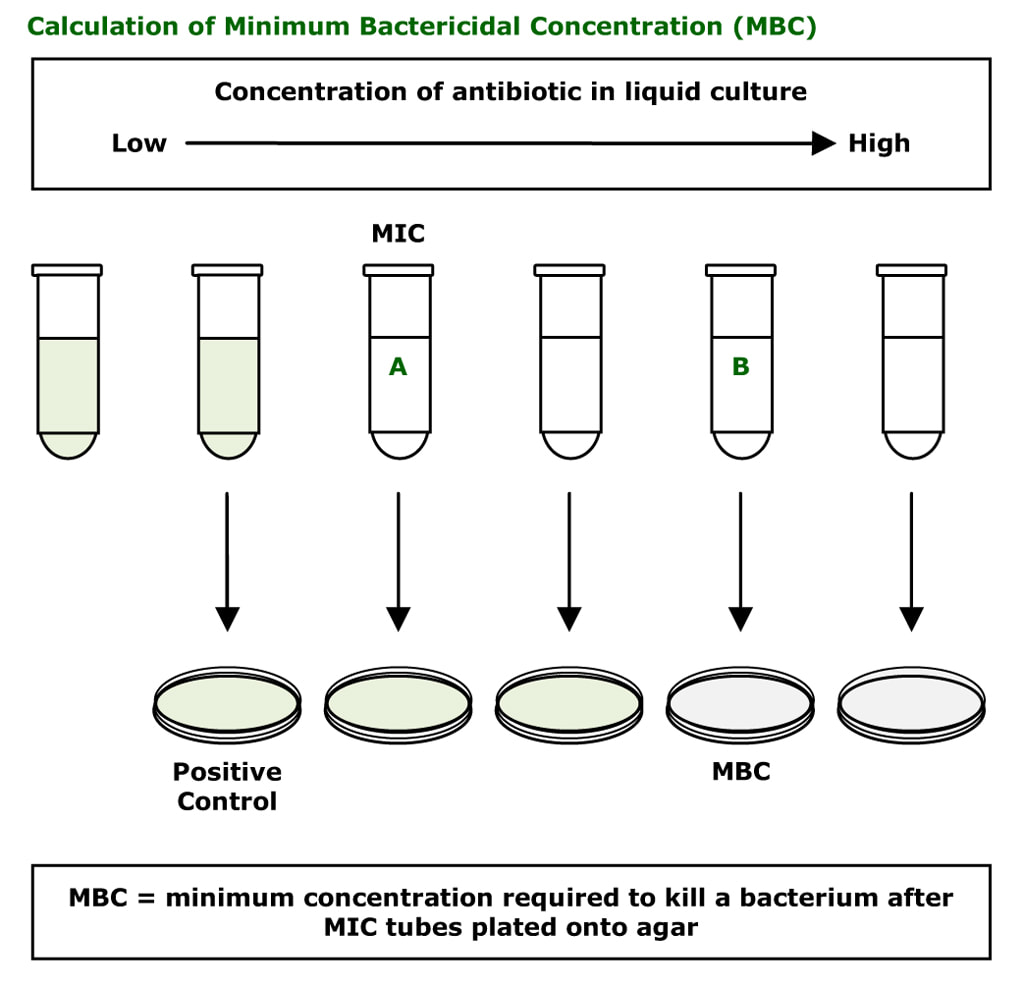
Minimum Bactericidal Concentration (MBC)
The MBC is the least amount of antibiotic required to kill a bacterium. It is very rarely performed. It is difficult to do and labour intensive. Different dilutions of antibiotic are prepared in liquid culture media from low concentration to high concentration. The bacterium is then inoculated into these tubes. After 24-48 hours the tubes where the bacterium is growing become cloudy (green tubes in the diagram below); some tubes show no bacterial growth (clear tubes in the diagram below). This test allows the laboratory to initially determine the MIC (the lowest concentration of antibiotic required to prevent a bacterium from multiplying). The first clear tube shows inhibition of growth and corresponds to the MIC. The MBC is determined by plating out the liquid cultures to agar. The first cloudy tube is known to have the bacterium growing and is used as a positive control, while the clear tubes have either inhibited or killed bacteria in them. The agar does not contain antibiotic therefore any living bacteria will now not be inhibited and start to grow (e.g. tube A). Tube B is the MBC; the bacterium in the tube has not grown on the agar because it has been killed by the concentration of antibiotic that was in tube B.
MBCs are only very rarely necessary, and only when you have a particularly unusual bacterium that will require cidal levels of antibiotic and you are unable to predict what these concentrations might be from published data… e.g. the infection is just too rare! This is a really time consuming tricky thing to do… it’s not normal lab work… don’t ask for it!!!
If you get a result that implies you cannot kill the bacterium with a single antibiotic agent, HECK what do you do then!! PANIC… Don’t panic. All is not lost. There is another level of antimicrobial susceptibility testing (this is levitation?) that can be done if you do an MBC and get this result. However I can count on 1 hand the number of times I have had to do this in the last 20 years; it’s the serum-cidal level! Dom dom dommmm…
Serum-cidal levels
When you do a serum-cidal level you are essentially looking to see whether multiple antibiotics, in combination with the patient’s immune system, are capable of killing the bacterium. Sounds tricky? It is…
In this situation the patient is usually on a combination of antibiotics that are expected to work synergistically with each other, that is the sum of their activity is greater than their parts. We often do this for complex infections such as endocarditis where we might use something like Teicoplanin, Gentamicin and Rifampicin together.
The problem with lab antibiotic susceptibility tests like disc diffusion, MIC or MBC is that they only test single antibiotic/bacterium combinations. What do you do if you want to work out whether a number of antibiotics working together, when combined with the patient’s immune system, will kill the bug?
Say our endocarditis patient is given Teicoplanin as the main antibiotic and Gentamicin and Rifampicin as adjuncts to “help” assist the Teicoplanin. In this situation you give the patient the antibiotics as “normal” (depending on frequency of dosing!), and THEN when you are about to give the next main antibiotic e.g. Teicoplanin, you take a blood sample.
The laboratory then separates off the serum (the clear stuff) and uses the serum containing antibiotics INSTEAD OF USING PURE ANTIBIOTIC in the MBC test. The laboratory tests doubling dilutions, each time decreasing the concentration of serum containing antibiotic against the bacterium in the tubes. At the end of the test, the laboratory have a titre of serum required to kill the bacterium e.g. NEAT, 1:2, 1:4, 1:8, 1:16 etc. The serum is said to be cidal if the titre required to kill the bacterium is 1:8 or more dilute (the cidal activity is able to be diluted more and still have an effect) i.e. 1:16 is good, 1:4 or above is not good!
As you can imagine, we don’t do serum-cidal levels very often. I have mainly used them when I have been treating really unusual bacteria in patients where I think the tolerance is extremely high, the patient is immunocompromised or where I keep getting positive cultures despite the antibiotic treatment! In these situations I need to know why the treatment is failing.
So, for our patient “we have done” an MBC (a rather talented Clinical Scientist…not me, I just don’t have the patience and subtlety to get them right!) and shown that we can get adequate serum levels which will overcome the bacterial tolerance… which is a relief, because I really wouldn’t like to have to ask my rather talented Clinical Scientist to levitate and do serum-cidal levels… and nor would she!!

 RSS Feed
RSS Feed
