So, was this an early Christmas present, an opportunity to hide the data in the Christmas furore or did Brexit just swamp our news channels. Let’s have a look and see….
For their vaccine trial Pfizer recruited 43,548 volunteers from 152 sites Worldwide (130 in the USA, 1 in Argentina, 2 in Brazil, 4 in South Africa, 6 in Germany and 9 in Turkey). 21,720 volunteers received the vaccine and 21,728 received a placebo. At the time of presenting their paper there is 2 months of data, from a trial that is supposed to last 2 years. In addition, although the vaccine was given to patients with Human Immunodeficiency Virus (HIV) infection that data has been excluded from the current report; but we are told it will be reported separately which is in fact a good idea as it may make the results in non-HIV patients difficult to interpret.
In terms of the definition of a case of Covid-19 this was defined according to the Food and Drug Administration criteria of fever, cough, shortness of breath, loss of taste or smell, muscle pains, diarrhoea or vomiting PLUS a positive respiratory sample tested for SARS CoV2 by PCR. This is a broader case definition than the main definition used in the Oxford study, but the Oxford study did expand their data to include any symptoms as well as asymptomatic infection… this study does not have any data for asymptomatic infection.
So, what have we learned?
The good
From the data presented the Pfizer vaccine looks very good at preventing symptomatic infection with SARS CoV2, and I mean VERY GOOD, preventing 95% of Covid-19 infections. Some examples of how effective other vaccines in routine use are:
- Rotavirus – 85%
- Streptococcus pneumoniae – 60-70%
- Haemophilus influenzae type b (Hib) – 85%
- Measles (MMR) – 97%
- Mumps (MMR) – 88%
So it’s not impossible that the Pfizer vaccine is as good as the “press release” data suggests it is but I am a little worried when I compare it against these other vaccine percentages; if it looks too good to be true then I start to doubt the validity of the result, but time will tell if it really is this good… let’s hope so. Even if it’s not, it should still hopefully be “good enough” to help to control the pandemic.
The Pfizer vaccine is said to be very good at preventing severe Covid-19 infection, but this is not 100%; 1 out of 8 Covid-19 cases in the vaccine arm of the study still got severe Covid-19. The numbers are small but the percentage of severe Covid-19 amongst the cases in the two arms of the study are actually higher in the vaccine arm (12.5% vs 5.6%) but I suspect this may be a quirk of the small numbers as the normal quoted rate of severe Covid-19 is 10-15% so small numbers may give either a falsely high or low figure in comparison to the millions of cases that have occurred Worldwide.
The vaccine and placebo groups were well matched for the demographics presented, but there are some key areas not covered (see the not so good below). An area which is particularly good is the spread of different ethnic groups as well as a high percentage (35%) of obese volunteers… I never thought I would say obese was good, but it does mean that the groups are representative of the UK population and other developed countries. Yep that’s all I have to say on the good!
The not so good
Adverse events were more common with the vaccine than placebo (26.7% vs 12.2%) with 0.2% of vaccine recipients having reactions leading to them withdrawing from the trial (that’s 2 in 1,000 recipients). I don’t know if this is a high rate of drop out but it seems pretty high to me when you think about the perceived danger of Covid-19 and the protection recipients would have from a vaccine… you’d think people would want to stay in the study.
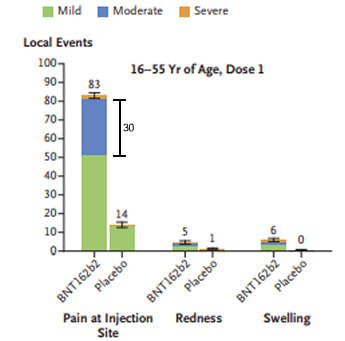
The local injection site side effect rate from the Pfizer vaccine is extremely high. Approximately 80% of 16-55 year olds had local side effects, and 70% of >55 year olds, compared to 12% and 8% with the placebo. Pfizer say the reactions were “mostly mild or moderate” consisting mainly of pain and swelling but don’t state what the percentages of moderate versus mild are… why lump all these into one group? Looking at the Figures presented in stacked column graphic format I would estimate that about 30% of 16-55 year olds and 15% of >55 year olds had moderate or severe local reactions whereas there were none in the placebo group. Guess what? What these reactions actually were wasn’t included either. In fact this seems to be very much played down in the paper and I believe that’s wrong. However, to put these “local reactions” into context mild pain is a common feature of many vaccines; 80% with the Human papillomavirus vaccine (HPV) and 60% with the Oxford vaccine.
- Fever – 16% (0%)
- Fatigue – 59% (23%)
- Headache – 52% (24%)
- Chills – 35% (4%)
- Muscle pain – 37% (8%)
- Joint pain – 22% (5%)
These rates are high, especially when compared with systemic side effects like this occurring in only up to 5-15% of HPV and Oxford vaccine recipients. The rate of side effects in the placebo arm does also look quite high, which may be due to a suggestive bias in the way volunteers were asked about symptoms, “oh do you have a headache?” , the answer is often “yes”. We also still do not know how long these side effects last as only fever and chills are mentioned in the paper as lasting a few days; what about muscle and joint pains? I have heard anecdotal evidence that it can be weeks!?! If they are short term and only last a few days this may be acceptable, but if they last for weeks or months and adversely affect the life of the “previously feeling good” vaccinated person then maybe they’re not.
I would like to see more demographic data; in particular I would like to have a better idea of what the risk of contracting SARS CoV2 infection was in the vaccine and placebo groups, a healthcare worker is clearly higher risk than a homeworker in finance. I would expect to know the number of high-risk professions (e.g. health care workers as well as other key workers) in each group, be it the vaccine or placebo, as if this is not balanced the difference between the two groups could just be about the level of exposure to the virus rather than protection from the vaccine. Added to this I’d have liked to see included knowing whether the groups were balanced by the countries in which the vaccine was tested and therefore if the exposure to SARS CoV2 due to location would be the same, but it’s missing. Also the detail about the ages of volunteers is sparse, with only the average age and range being given, age is a key factor in terms of risk of severe disease as well as potential exposure through working; really it should be presented in more detail. I am worried that this information is missing as it is fundamental and I believe it should have been picked up by the Editor or reviewers at the NEJM, to my mind it either wasn’t collected which would be poor practice, or it is not balanced and therefore not presented, either way it is concerning.
The Pfizer vaccine trial does not appear to have been set up to detect asymptomatic infection and so we don’t know if the vaccine prevents asymptomatic infection and therefore asymptomatic transmission. This again looks like a BIG oversight! Surely anybody producing a vaccine would want to be able to show that it reduces carriage rates. So did Pfizer, a pharmaceutical giant, fail to set up the trial correctly, or does the data show that the vaccine doesn’t prevent carriage and they just don’t want to admit that?
Added to my mounting “another area of concern” is that Pfizer had complete control of all the analysis and write up, the study researchers were not involved at all; “Pfizer was responsible for the design and conduct of the trial, data collection, data analysis, data interpretation, and the writing of the manuscript” (Safety and Efficacy of the BNT162b2 mRNA Covid-19 Vaccine, NEJM 2020). This contrasts with the Oxford study where the opposite was true, and the researchers had complete control and Astra Zeneca did not influence the publication at all. I find this a little disturbing as it introduces that OLD spectre of bias again with Pfizer only “telling us what they want us to know” and potentially holding back any bad news. OK I might be being a bit paranoid but why else do it this way? Tut Tut Tut, it doesn’t give me fuzzy warm feelings of trust Pfizer!
Observation from the data but not part of the trial
The figure in the paper that shows the number of cases in the vaccine and placebos groups is actually split into two; a group with no evidence of previous infection with SARS CoV2 and a group that also includes some volunteers with evidence of previous infection with SARS CoV2. Their purpose of the figure is to show the effectiveness of the vaccine, but look at it in another way and this table shows that in the unvaccinated group, 7 cases of Covid19 occurred in 1,847 people who had previously been infected. Let’s rephrase that: the rate of reinfection in those who have been infected within the last 6 months was 0.4%... which means that just because you’ve had the infection before doesn’t mean you can’t get it again! YIKES! That is concerning epidemiologically and it also has wider implications on transmission and the length of this Saga.
So, what comes next?
In the Pfizer vaccine study we have 2 months of preliminary data about the safety and efficacy of a Covid-19 vaccine, and in the proposed 2 year time scale of the study we will have the same amount of safety and efficacy data for the vaccine compared to placebo… WRONG! It is very likely that this trial will now end. YEP it’s to end after just 2 months! WHY I hear you cry? Well, it is now deemed “unethical” to leave the volunteers without protection from the vaccine. To quote Pfizer “although this study was designed to follow participants for safety and efficacy for 2 years after the second dose, given the high vaccine efficacy, ethical and practical barriers prevent following placebo recipients for 2 years without offering active immunization”. Whilst I understand the ethical dilemma, I think this is wrong, surely this problem was covered in the “consent procedure” for this clinical trial and the volunteers were aware that they were not be eligible for the vaccine before the end of the trial? We now will not know the difference between the vaccine and placebo over more than a 2-6 month period, and that doesn’t provide me with assurance that this is “all safe” or “long lasting”; if it was only short term immunity would it actually be cost effective!? Remember those deep freezers and profit margin price tags.
Another worrying development is that the UK’s Department of Health has decided to go against Pfizer’s dosing recommendations of 2 doses 3 weeks apart and plough ahead and give the doses 12 weeks apart instead. The reason (excuse!?) for doing this is to try and vaccinate as many people as possible with at least 1 dose. The early data from Pfizer showed that at 7 days after the first dose of vaccine 70% of people had detectable antibodies which might suggest 70% of recipients were protected BUT remember this means 30% of people did not have protection. It also doesn’t mean that a booster given at 12 weeks will be as effective as that given at 3 weeks, and it doesn’t mean that 1 dose of vaccine will protect people for 12 weeks… these are all unknowns! Surely, it’s better to protect 1 million people properly than not know what is going on in 2 million people? It sounds like a political “aren’t we great sound bite” to me!
I really do not see the value in doing this. I may still have concerns about the Pfizer vaccine, but if we are to use it, let’s use it properly and as the manufacturer recommends. Not only does this decision mean we have “no idea at all” how effective the vaccine is going to be BUT we also now cannot hold Pfizer to account if anything goes wrong as they can just say we didn’t use it properly…. CRAZY!
Hey, but at least we now have data on the Pfizer vaccine and you can all read the paper for yourselves and see whether you agree or not. GOSH, this sounds like the basis of peer review science and informed consent… and that IS a good thing.
Happy New Year… let’s hope it’s a better one than last year….

 RSS Feed
RSS Feed
